Six key trends and insights drug developers need to know to succeed in 2026
Insights from the 2026 Pulse Survey of global drug development leaders

The 2026 landscape at a glance: Navigating uncertainty while accelerating progress
The drug development landscape in 2026 is defined by heightened uncertainty, rising costs, increasing trial complexity and a changing regulatory landscape. Despite the barriers, there is also unprecedented opportunity.
To better understand how sponsors are responding to these and other challenges, the PPD™ clinical research business of Thermo Fisher Scientific surveyed 150 senior leaders across biopharma and biotech organizations worldwide. This fourth edition of The Pulse survey reveals how drug developers are navigating shifts by balancing scientific ambition with operational realities, adopting new technologies, and refining strategies to remain resilient and competitive.
The findings underscore a clear tension: sponsors are under pressure to move faster and spend smarter, even as the regulatory landscape shifts and cost and complexity intensify. The findings also highlight that leveraging technologies and innovations, particularly in trial design, artificial intelligence (AI) and patient-focused strategies, are becoming critical levers for success.
What drug developers need to know to succeed in 2026
1. Rising costs and trial complexity remain the industry’s biggest hurdles
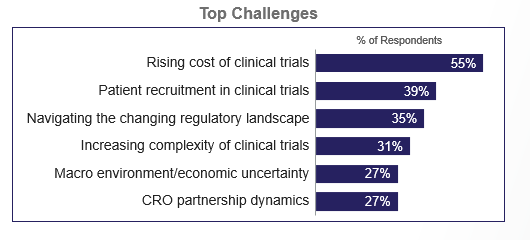
The rising cost of clinical trials continues to be the number one hurdle drug developers face, followed by patient recruitment challenges and a rapidly changing regulatory landscape. This year, regulatory changes have surged in importance, reflecting growing uncertainty around global policy shifts, U.S. health care reforms and evolving regulatory agency expectations.
Nearly half of the organizations surveyed report that their clinical development timelines have lengthened compared with two years ago. The primary drivers are familiar (complex protocols, challenges enrolling and retaining patients, and regulatory requirements) but have become increasingly intertwined.
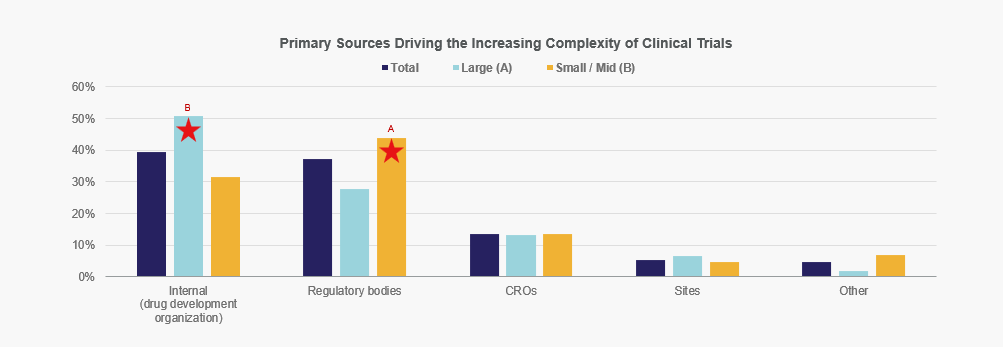
Overall, trial complexity is driven equally by internal factors and regulatory requirements, but Large companies put more weight on internal considerations while Small/Mid-size organizations assign greater weight to regulatory bodies.
Letters indicate statistically significant difference between groups at the 90% confidence level
Base = all respondents. Total: n=150; Large: n=61, Small/Mid: n=89
While more than 70% of sponsors believe study protocols are more complex than necessary, more than half also agree that this complexity is required to meet scientific and regulatory objectives. This paradox underscores the difficult trade-offs, between efficiency and rigor, that sponsors face.
To counter, most organizations are actively deploying strategies to reduce timelines, including innovative trial designs, increased outsourcing and greater use of AI and machine learning (ML).
2. Innovative trial designs move from aspiration to necessity
Innovative trial designs have emerged as one of the most important tools for streamlining execution and overcoming cost and timeline pressures. Sponsors are increasingly using innovative methods, including adaptive designs, basket trials, and Bayesian statistical models to streamline execution and reduce sample sizes, particularly in early-phase studies.
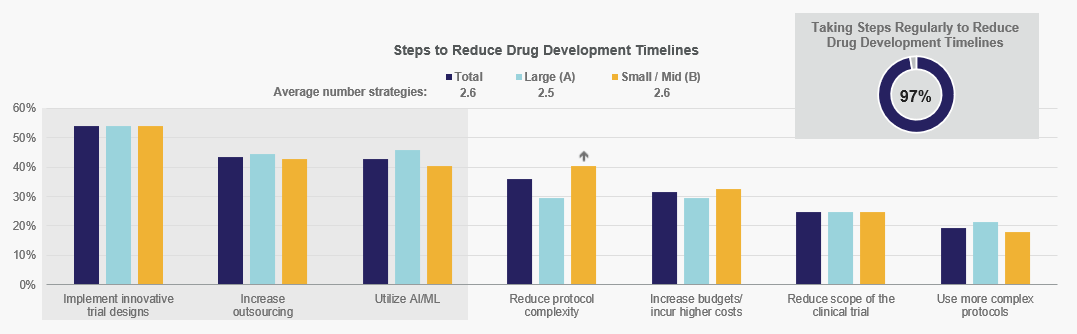
Implementing innovative trial designs is the most common tactic for reducing drug development timelines for both Large and Small/Mid-size companies, followed by increasing outsourcing and utilizing AI/ML. Directionally, Small/Mid-size sponsors are more likely to reduce protocol complexity to shorten timelines than their Large company counterparts.
No statistically significant differences between groups at the 90% confidence level
Base = all respondents: n=150; Large: n=61, Small/Mid: n=89
Many organizations report using expanded Phase I designs to generate early proof-of-concept or leveraging surrogate endpoints for faster readouts. These approaches enable teams to make earlier go/no-go decisions, skip development phases where possible and maximize data yielded from each study.
While innovative trial design is embraced across company sizes, small and mid-sized organizations are especially reliant on it to preserve capital, accelerate timelines and increase asset value in an uncertain funding environment.
3. Patient recruitment and experience are strategic priorities, but progress is uneven
Patient recruitment remains one of the most persistent challenges in drug development. In response, sponsors’ top patient-related goals in 2026 are increasing overall enrollment, improving retention and removing barriers to participation.
Organizations are investing in a wide range of patient-focused strategies, including patient-friendly technologies, clearer communication, shorter and fewer study visits, and stronger relationships with sites and advocacy groups. However, three out of four sponsors say they have achieved only limited success so far, indicating significant room for improvement.
Encouragingly, nearly all sponsors now track patient diversity, and many are seeing progress. Small and mid-sized companies have made notable gains in diverse enrollment compared with previous years, reaching parity with large organizations. Still, for roughly one-quarter of respondents, it is too early to determine whether diversity initiatives are delivering sustained results.
4. AI and ML are widely adopted, yet still evolving
AI and ML are now considered mainstream in drug development. Virtually all surveyed organizations use AI/ML in at least one area, with the average company leveraging these tools across 13-14 functional areas.
The most common applications include operations, data analysis, drug discovery and design, predictive insights, medical and scientific writing, and market analysis. Sponsors report tangible benefits, like enhanced data analysis, more accurate predictive modeling and improved biomarker identification.
While the full impact of AI/ML is still coming into focus, it is already helping organizations with mature programs shorten their development timelines. However, many also report increased costs and added trial complexity, reflecting the requisite investment and integration challenges that accompany advanced technologies. In addition, key barriers to broader adoption exist, including trust in AI outputs, integration with existing systems, regulatory uncertainty and limited internal expertise.
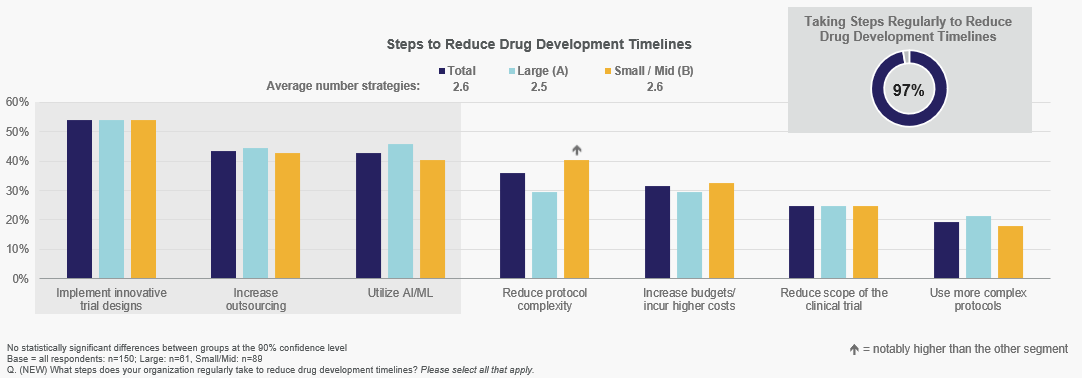
Implementing innovative trial designs is the most common tactic for reducing drug development timelines for both Large and Small/Mid-size companies, followed by increasing outsourcing and utilizing AI/ML.
Directionally, Small/Mid-size sponsors are more likely to reduce protocol complexity to shorten timelines than their Large company counterparts.
5. Outsourcing strategies continue to shift toward flexibility
Outsourcing remains a core component of drug development strategy, but how sponsors outsource is evolving. Full-service outsourcing (FSO) continues to account for roughly one-third of clinical development work, while functional service provider (FSP) and mixed models each represent about a quarter.
Large organizations are more likely to favor FSP models, reflecting efforts to maintain strategic control while scaling capabilities. Small and mid-sized companies, by contrast, rely more heavily on FSO models to compensate for leaner internal teams.
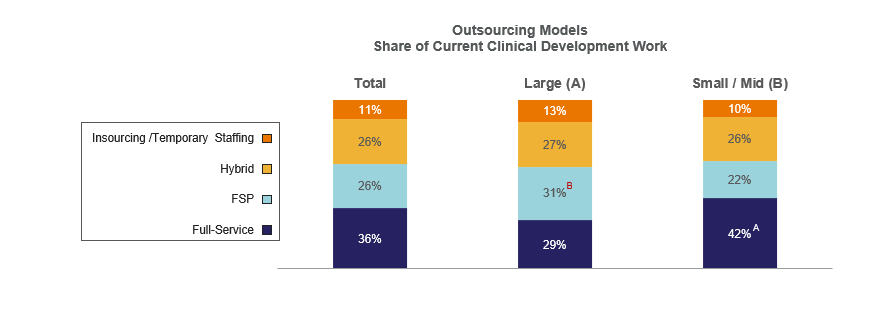
The full-service model is used for about one-third of current clinical development work, while FSP and Hybrid models are each used for about one-fourth.
Participants in Large enterprises use FSP more than their counterparts in the Small/Mid-size segment, while Small/Mid-size enterprises are more apt to use full-service outsourcing.
Letters indicate statistically significant difference between groups at the 90% confidence level
Base = all respondents; Total: n=150; Large: n=61, Small/Mid: n=89
Across the industry, clinical laboratory and diagnostic services and patient recruitment are the activities most likely to be outsourced over the next two years. Many sponsors are also rationalizing their vendor portfolios, seeking fewer, more strategic partnerships to improve efficiency and manage costs.
6. Uncertainty shapes strategy and decision-making
Perhaps the most defining theme of the 2026 Pulse survey is uncertainty. While 60% of respondents believe the industry is moving in the right direction, a substantial minority remain concerned about its trajectory. Macroeconomic pressures, geopolitical instability and health care policy changes (particularly in the U.S.) are influencing portfolio decisions and risk tolerance.
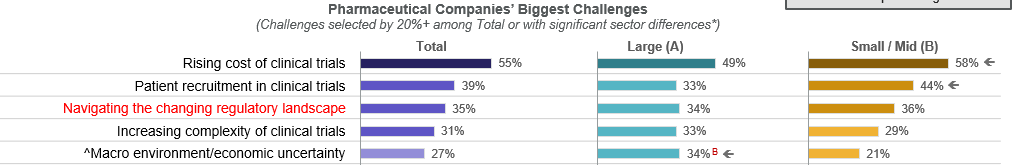
Regardless of size, the top clinical challenges for drug developers are the rising cost of clinical trials, patient recruitment, the regulatory landscape, and increasing trial complexity.
Significantly more Large companies are challenged by macro environment/economic uncertainty and feasibility/site selection, while lack of funding is a bigger problem for more Small/Mid-size organizations.
Letters indicate statistically significant difference between groups at the 90% confidence level
Base = all respondents: Total: n=150; Large: n=61, Small/Mid: n=89
As such, sponsors are prioritizing strategies that emphasize resilience: maximizing asset value, reprioritizing pipelines, managing costs and reducing operational complexity. Trust and collaboration with contract research organization (CRO) partners are also under scrutiny, as sponsors seek greater transparency and alignment in an increasingly high-stakes environment.
Success in 2026 requires agility, innovation and focus
Biopharma and biotech companies remain challenged by heightened industry uncertainty, ever-increasing trial complexity and costs, and navigating fluctuations in the regulatory landscape. As the industry continues to undergo these transformational shifts, success in drug development depends on a sponsor’s ability to adapt. Rising costs, complex protocols and regulatory uncertainty aren’t unlikely to disappear, but sponsors aren’t standing still. By embracing innovative trial designs, investing in AI and ML tools and other technologies, sharpening patient-focused strategies, and adopting more flexible outsourcing models, drug developers are finding new ways to advance and thrive despite the shifting landscape.