
PPD Laboratory Services, Bioanalytical Lab
Bring your compound to market quickly and efficiently with an experienced partner
In today’s ever-evolving scientific landscape, it’s important for drug developers to stay ahead of and leverage the latest tools and techniques. It is essential to collaborate with a reliable partner who possesses scientific expertise and incorporates cutting-edge technology into your bioanalytical studies, from non-proprietary assays and biosimilar to cell and gene therapies.
Over the past 30 years, our bioanalytical lab has helped bring thousands of compounds to market by employing the most technologically advanced systems to develop assays and applying them to clinical trial samples across every stage of drug development.
PPD™ Laboratory services deliver fast, precise and reliable results while ensuring regulatory compliance. With us, you can confidently embrace scientific advancements and achieve your bioanalytical goals.
Strategies to Improve Comparability Assessments Across Bioanalytical Data for Biosimilar Studies
Experience and capabilities
Scientists across bioanalytical, biomarker and vaccine specialties
Validated methods
Square feet of lab space globally
We continuously seek innovative ways to push the boundaries of the bioanalytical laboratory services industry, always aiming to provide you with the best possible results. We have significant bioanalytical experience working with:
- Small molecules
- Biologics
- Vaccines
- Biomarkers
- Cell and gene therapies
- Biosimilar
We are at the forefront of biologics LC-MS innovation, and our clients benefit from our advancements in analyte isolation, detection and measurement. We have extensive experience working with complex molecules such as oligonucleotides, monoclonal antibodies and protein biotherapeutics.
Supported 25 different biosimilar products (including 11 of the first 12 ever approved)
With our extensive inventory of state-of-the-art instrumentation, we have the capability to address all your bioanalytical laboratory testing needs under one roof. This means you can rely on us as your trusted single provider, streamlining the process and ensuring efficient and accurate results. Our capabilities span a wide variety of technologies and therapeutic areas:
- Immunochemistry
- Chromatography
- Cell-based assays
- Flow cytometry
- Molecular genomics
- Biostatistics
- ADC development
Our teams analyze over 90,000 samples across 250+ different protocols each month
Our team also has more than 30 years of experience in bioanalytical veterinary testing. Our bioanalytical lab’s veterinary pharmaceutical services include customized/proprietary assays, discovery analysis studies for final product specifications, and sample analysis planning, which enable us to streamline the pre-concurrence process.
Explore our flagship laboratory in Richmond, Virginia
People-driven approach
Our people are at the heart of our bioanalytical solutions. Our experienced scientists specialize in bioanalytical testing for drug and biologics development. With an experienced lab management team, we bring unmatched expertise and longevity to every project.
Our bioanalytical lab leverages the expertise of our leaders, managers and scientists to meet your requirements for accurate, timely and high-quality results. Our clients benefit from:
- Our technical, cross-discipline know-how and effective solutions to complex analytical challenges.
- Consultation and guidance on procedures/methods to obtain the best data to meet specific program goals.
- Proven track record of regulatory compliance in GLP and GCLP, as well as an in-depth understanding of current regulatory guidance.
- Full biostatistical support for assay development, qualification and validation, and cut-point analysis for biologics.
- Our project managers, with 10+ years of extensive bioanalytical experience, who support your organization. These specialists serve as a dedicated liaison between your team and the rest of the PPD team, ensuring seamless communication and collaboration throughout the project.

Integrated laboratories
With a large bioanalytical lab spanning 430,000 square feet and a skilled team, we handle your entire protocol simultaneously, running tests in parallel for maximum efficiency. This approach ensures timely results, accelerating informed decisions and reducing both time and costs.
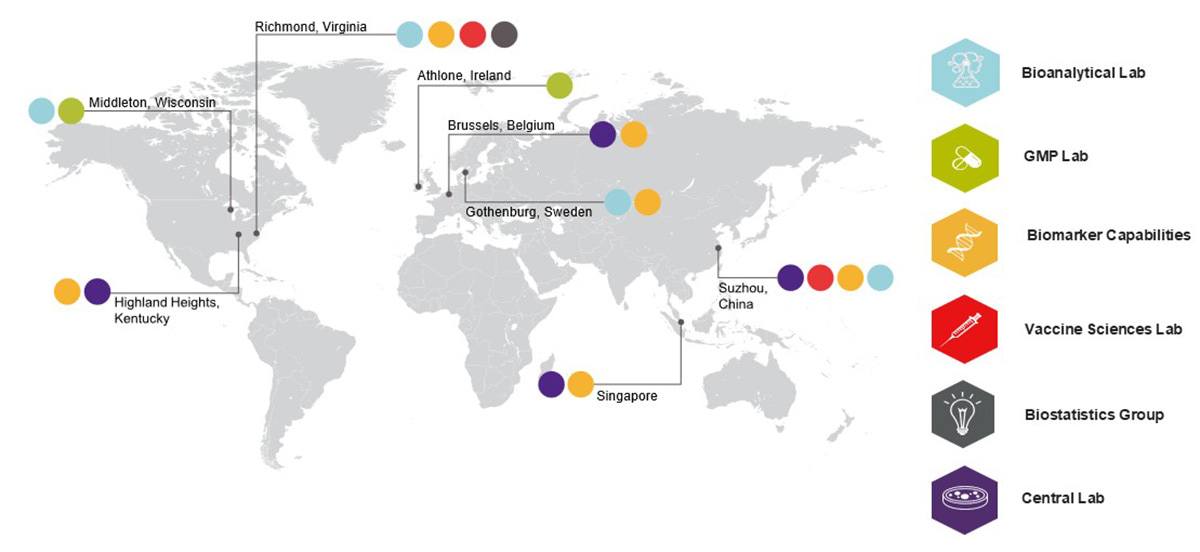
Projects that utilize our central lab benefit from the integration of data through our award-winning PreclarusTM lab solution. This solution offers additional levels of visibility for key project metrics and provides real-time access to select data sets.
When working with our bioanalytical lab, customers leverage integration of both our vaccine sciences lab and our biomarker lab for their projects. This approach leverages our extensive bioanalytical experience to enhance the expertise of the vaccine sciences and biomarker teams.
Our extensive automation capabilities in robotic liquid handling, project management, process improvement and e-data ensure that your samples flow through our bioanalytical laboratory faster and more effectively.
Learn more about our vaccine sciences lab and biomarker capabilities.
Our experts help you navigate and expedite the complex regulatory process
With rapidly evolving global regulatory guidelines, it is essential that you partner with an experienced and knowledgeable team that can navigate these complexities with ease. Our bioanalytical lab has successfully completed more than 80 regulatory inspections over the past three decades, including audits of multiple programs across lab disciplines. As a result, we can quickly anticipate and overcome potential regulatory challenges due to evolving expectations for bioanalytical clinical data sets that could pose obstacles to regulatory approvals.
successful regulatory inspections over the past three decades
Tech Talks:
PPD Laboratory Services, Bioanalytical Lab
Watch as experts from our bioanalytical, biomarker and vaccine sciences labs take you through trending topics of the bioanalytical landscape ranging from CAR-T cell enumeration to vaccine immunogenicity testing.
Related resources
Learn more about PPD Laboratory services bioanalytical lab.
To request a proposal or contact your local business development representative, please complete the form below.

