PPD Functional Service Partnership (FSP) Pharmacovigilance Solutions
Flexible solutions underscoring a commitment to patient safety
Pharmacovigilance complexity continues to increase, driven by evolving global regulations, rising adverse event volumes and increasingly distributed clinical trials. As development programs expand, organizations must ensure their pharmacovigilance operations adapt quickly to maintain compliance, manage data efficiently and support critical timelines.
PPD™ Functional Service Partnership (FSP) Pharmacovigilance solutions provide confidence as you scale. By combining advanced technology, including AI-enabled case intake and automation, with deep global expertise, we enhance operational efficiency while supporting high-quality, compliant safety reporting. Our flexible FSP solutions complement your existing capabilities to optimize your resources, manage costs and ensure on-time delivery of therapies (whether in development or in market) to patients who need them.
A full range of capabilities and unmatched expertise
PPD FSP Pharmacovigilance solutions empower biopharma and biotech companies to confidently manage the growing complexity of safety data through a flexible, partnership-driven model. By combining the advanced technologies of our PPD SafetyNet Suite—with deep therapeutic and functional expertise, we help streamline case processing, enhance data quality, and maintain global compliance.

The ease of a single, flexible and capable partner
By choosing an experienced, industry-leading functional service provider for your pharmacovigilance solutions, you gain a collaborator with a proven track record of stable, high-quality services and the convenience and ease of a single global partner.
With over 30 years of pharmacovigilance and FSP expertise, our solutions support all of your clinical trial and post-marketing service needs and bring robust therapeutic area experience and global expertise from more than 35 countries.
- Case processing
- Artificial intelligence (AI) driven automated case intake
- Safety reporting
- Global literature services
- Safety science including signal management
- Safety writing
- EU/UK specialty services
- Local pharmacovigilance services
- Pharmacovigilance agreement (PVA) management
- Medical safety evaluation and risk mitigation (M-SERM) physicians
- Pharmacovigilance consulting
- Pharmacovigilance technology consulting
- Medical good clinical practice (GCP)services
Breadth and depth of pharmacovigilance expertise
Experience matters, which is why over 1,100 biopharmaceutical, biotech and medical device organizations have turned to PPD FSP Pharmacovigilance solutions over the years for pharmacovigilance support.
years of experience
companies supported since 1997
pharmacovigilance experts in 35 countries
endpoint dossiers adjudicated
ICSR cycles processed annually
recipients sent individual case safety reports (ICSRs) each year
on-time safety reporting compliance rate
Ensuring effective pharmacovigilance regulatory intelligence with FSP partnership
Get the right experience, knowledge and expertise from top-tier professionals
To manage your complex pharmacovigilance efforts, you need professional know-how. That’s why you should choose a contract research organization (CRO) with a deep bench of more than 1,400 pharmacovigilance experts, comprising highly trained physicians, pharmacists, nurses, medical scientists and health care professionals.
But expertise isn’t enough – you need stability. Our clinical research team’s culture of professional development and learning fosters commitment that, in turn, delivers business continuity and the confidence that your programs are supported by dedicated professionals who keep their fingers on the pulse of the industry and apply the right experience, knowledge and expertise throughout your product’s life cycle.
To keep employees on top of ever-evolving regulations and technology changes, we offer award-winning employee development programs. Trainings include corporate and departmental training, mentoring and shadowing, as well as training developed specifically for pharmacovigilance client programs. Our professional development and learning culture make us a great company to work for, thereby increasing pharmacovigilance employee engagement and retention (average tenure is 6 years for senior & principal level staff and 8.5 years for manager level and above). Our industry-leading retention delivers business continuity and the confidence of knowing that your pharmacovigilance system is supported by quality professionals who apply the right experience, knowledge, and expertise.

Technology to enhance patient safety monitoring
As drug development activity increases, drug developers face rising individual case safety report (ICSR) volumes, increasingly complex reporting requirements and diverse report formats. To successfully navigate the changing environment, biotech and biopharma companies need pharmacovigilance solutions that leverage new technologies, analytics, process improvements and automation to more easily scale capacity, improve consistency, and shorten cycle times without compromising quality and compliance, reducing the pharmacovigilance cost burden.
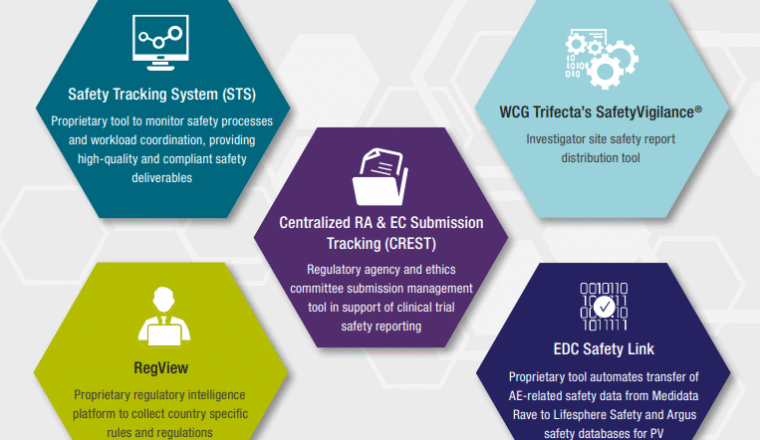
We are continuously innovating and advancing the systems, processes and technologies that assure excellence in your patient safety monitoring. To unlock efficiencies, we offer a range of bespoke pharmacovigilance technology capabilities, tools and systems.
PPD SafetyNet Suite is a comprehensive group of pharmacovigilance (PV) drug safety technology platforms that support every phase of pharmacovigilance, improving consistency and quality, driving down costs and giving you easy access to your data. Our tools bring you greater efficiency, flexibility and insights.
- PPD SafetyNet Signal
- PPD SafetyNet Tracking
- PPD SafetyNet Dashboard
- PPD SafetyNet Intelligence
- PPD SafetyNet Intake
- PPD SafetyNet Processing
- PPD SafetyNet Submissions
- PPD SafetyNet Literature
Contact us
Ready to transform your approach to pharmacovigilance? Connect with our team for customized approaches that provide resource flexibility, reliability and continuity.