
PPD Laboratory Services, Comprehensive Biomarker Solutions
A holistic approach to streamline biomarker development, enhancing your drug’s potential for commercial success
The drug development field faces numerous challenges, with low success rates being a major concern. Biomarker analysis plays a crucial role achieving success by determining the efficiency, effectiveness, and safety of a drug, enabling you to make informed decisions throughout the development process and ultimately accelerating it to the market.
A biomarker-driven approach enables risk mitigation throughout the drug development process, meaning:
- Clinical trials are twice as likely to succeed if drug development companies use them.
- The use of biomarkers triples the chance of a drug moving from Phase I trials to market.
Our biomarker solutions engage biotech and pharmaceutical companies early in the drug development process, using our comprehensive analysis of biomarkers of interest and custom assay development to enhance efficiency and synergy.
Delivering the right expertise and capabilities
By leveraging a diverse set of technologies, robust project management and dedicated expertise, your assay is supported throughout the drug development process globally.
Studies completed in the past five years
Different compounds supported
A seamless biomarker service experience
Our innovative approach aligns global lab capabilities and bioanalytical offerings to deliver cohesive and efficient biomarker services spanning the drug development timeline across several platforms.
Our biomarker services offering will make your experience seamless from method development to technology transfer. Whether you have a kit or a custom assay, exploratory or clinical sample analysis, our scientists optimize development and move your program through to global sample testing.
Method development
Verification and exploratory study support
Full method validation
Technology transfer globally
Integrated approach to meet your study needs
The integration of our biomarker capabilities into both our PPD™ Laboratory services Central and PPD™ Laboratory services Bioanalytical labs enables us to fully customize your biomarker development strategy and provide global coverage for studies of all sizes.
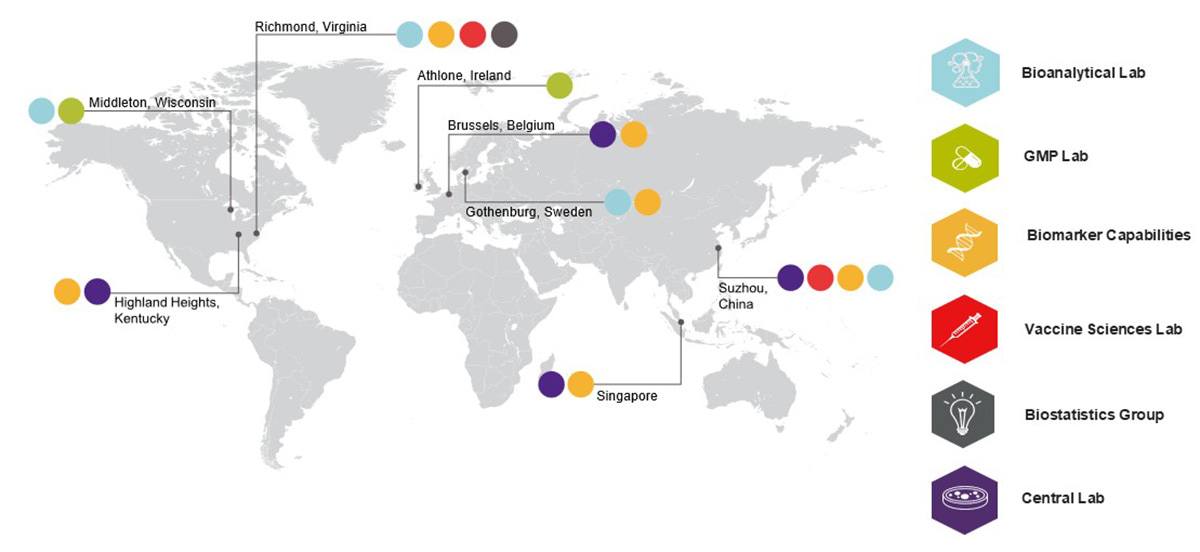
- Offers global coverage for studies of all sizes
- Allows the biomarker team to provide a customized development strategy for each project
- Leverages the operational experience of the PPD Bioanalytical and PPD Central labs
- Provides access to lab data in real time through our Preclarus™ LIMS
- Enables the biomarker lab to apply a tiered regulatory approach from fit-for-purpose characterization to GLP/GCLP to CAP/CLIA
- Provides a dedicated project manager to streamline communication between your team and our scientists, as well as sites, couriers and any third-party labs, and serves as your single point of contact
Learn more about our biomarker capabilities
To request a proposal or contact your local business development representative, please complete the form below.



