Archives: Resources
Optimizing site activation to accelerate clinical trials
Discover strategies to overcome clinical trial challenges in the biopharmaceutical industry.
Key considerations when selecting an FSP partner for regulatory publishing
Unlock the secrets to efficient regulatory publishing. Learn how to streamline your processes and ensure compliance and gain valuable insights to stay ahead in the regulatory landscape.
Leveraging remote-based regional hubs in FSP partnerships
Discover ways to rethink traditional staffing and find the best-suited and most cost-effective professionals — wherever they are.
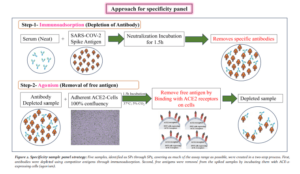
In Vitro Immunoadsorption and Antigen Agonism
Dive into our latest study to discover groundbreaking advancements in the assessment of COVID-19 vaccines.
Biosimilar development brochure
Accelerate clinical trials with our expertise in biosimilar development.
How digitized protocols accelerate clinical research
Discover how digitized protocols are transforming clinical research. Learn about the benefits of digital clinical trials, including improved efficiency, data accuracy, and faster timelines.
The 2025 State of FSP Outsourcing: Challenges, Trends and Opportunities, and the Future of FSP Strategies and Models
Discover how FSP and hybrid FSP/FSO models are helping sponsors better navigate clinical development outsourcing.
Real-world evidence studies – Diverse Data Designs
In close collaboration with sponsors, we identify and understand multi-stakeholder evidence needs across the product lifecycle.