Neurodegenerative Disease Research Expertise
Experience with neurodegenerative indication research studies
Global rates of neurodegenerative diseases — including Parkinson’s disease and dementias like Alzheimer’s disease —are increasing, and a significant gap remains between the clinical need for neurodegenerative treatments and the options available to patients and their caregivers.
To chart the best path forward for neurodegenerative disorder therapies, drug developers require partnership with a research team that has the operational and medical knowledge across indications and can leverage novel approaches, such as decentralized trials and clinical trial simulations.
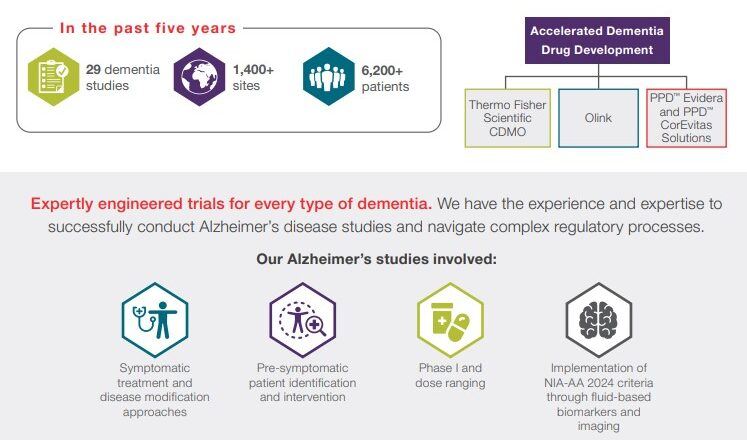
In the past five years, the PPD™ clinical research business of Thermo Fisher Scientific has performed more than 50 studies across a range of rare neuroscientific indications. This includes studies pursuing treatments for neurodegenerative diseases, including:
- Alzheimer’s disease (disease modifying and symptomatic)
- Dementia with Lewy bodies
- Frontotemporal lobe dementia
- Gene therapy
- Parkinson’s disease
- Radiopharmaceutical
- Single-ascending-dose (SAD) and multiple-ascending dose (MAD) trials

Solutions for neurodegenerative clinical trial sites and patient access
Gaining unique access to indication-specific sites for neurodegenerative clinical trials and a worldwide site network is critical to driving your therapy forward. Access to sites and site networks is just the beginning. These sites must prioritize ongoing support and training of health care professionals in the clinical trials space to ensure the delivery of industry-leading speed, skill, efficiency and quality benchmarks across neurodegenerative trials for all indications. By partnering with us, you’ll have access to the site and training services you need, including:
Site and training services include:
- Our wholly-owned clinical research unit in Orlando, Florida, which has extensive experience in neurodegenerative disorders, early development and SAD/MAD research
- SiteCoach, our training program to support health care practitioners at sites new to clinical research
Robust patient access solutions drive neurodegenerative disorder trial success
Success in neurodegenerative disorder trials depends on patient and caregiver participation. To reduce the individual burden for patients and their caregivers — and ensure patient enrollment and retention — drug developers should partner with a contract research organiztion (CRO) that provides extensive patient support services.
Our extensive patient support service offerings include:
- Enhanced diversity and collaboration with patient advocacy groups
- Helps ensure enrollment aligns with real-world disease incidence and regulatory guidance
- Increased access through minimized patient burden
- Home health care options, including partnering with remote staff
- Digital and hybrid clinical trial solutions and protocols leveraging mobile pages, wearables, telehealth and e-consent
- Proactive patient navigator services that offer reliable patient transportation and travel expense verification
- Flexible options for patient reimbursement in more than 150 currencies


Clinical trial design for Alzheimer’s disease research with ACE Simulator
Our scientists designed the Archimedes condition-event (ACE) simulator by building on widely used models for treating Alzheimer’s disease. The ACE simulator assesses interventions from the earliest stages (i.e., mild cognitive impairment or prodromal Alzheimer’s disease) through the most severe stages of the neurodegenerative disease, mapping the transition from normal cognitive function and helping optimize trial design.