
Regulatory Insights: ‘Right First Time’ Approach to Post-Authorization Strategy
Proactive post-authorization regulatory requirements intelligence is essential in the pharmaceutical industry. Learn about regulatory authority requirements.

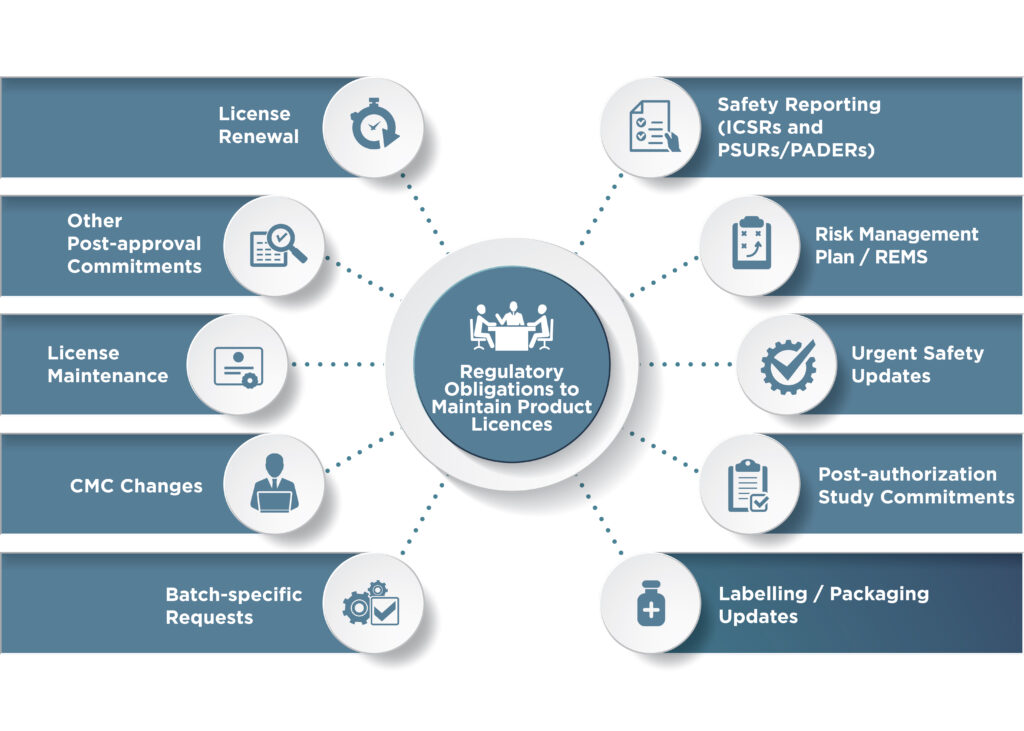
Throughout the product life cycle, license holders must navigate myriad evolving regulations to maintain regulatory compliance and monitor drug safety, typically across multiple global authorizations. In this post, Dushyant Kumar, senior manager of regulatory affairs, Shabana Bano, manager of regulatory affairs, and Archie Poole, associate director of pharmacovigilance, discuss the importance of regulatory intelligence in anticipating challenges and mitigating risks during post-authorization life cycle maintenance.
Understanding pharmaceutical post-authorization regulatory requirements
Around the globe, regulatory authority requirements across jurisdictions present significant challenges to the pharmaceutical industry in maintaining regulatory compliance and managing drug safety. Proactive regulatory intelligence is essential to identify legal obligations and for adoption of a “right first time” regulatory strategy to drive regulatory action.
Maintaining licenses
Terms of a product’s license may subsequently need to be changed after a medicine has been authorized for marketing. Reasons for such changes may include an update of product information leaflets, a change of site of batch release or extensions of the product’s shelf life, among others. Regulatory authority requirements and processes differ across countries, but similarities to streamline operations can be found that could benefit sponsor companies. For example, changes to product licenses are generally classified across jurisdictions as major or minor, distinguished based on their impact to the quality, safety and efficacy of the product and potential impact to public health.
In some jurisdictions, certain changes must be approved by regulatory authorities before they can be implemented. The timelines for these approvals may also vary depending on the type of changes being made. Awareness here is particularly important for chemistry, manufacture and controls (CMC) changes where coordination between regulatory, supply chain and manufacturing site personnel is often necessary to avoid interruption to supply. As an example, CMC changes in the U.S. can be submitted via an annual report (retrospective), changes being effected supplement (immediate), changes being effected in 30 days supplement (30-day review) or prior approval supplement (4-10 months).
Despite certain similarities across jurisdictions, global harmonization does not exist and the need for awareness of local post-authorizations requirements remains. For example, in Brazil some CMC changes are submitted via annual reports on a “do and tell” basis, whereas in Turkey some of these changes require Good Manufacturing Practice (GMP) certification before submission, a process that can take between one-and-a-half to two-and-a-half years. A manufacturing site transfer is considered a variation in some countries (e.g., China, Brazil and Singapore), but in others (e.g., Hong Kong) a new initial registration application is required. Regular, proactive regulatory intelligence is recommended to support early awareness of local requirements, regulatory planning and market compliance.
Labeling and safety changes
Accurate labeling and package leaflets are essential components for the safe and effective use of medicines and post-authorization compliance. Safety-related updates to the labeling can be prompted through various means. For example, authorities may spontaneously request the license holder to update information, or the license holder may initiate the process following internal review of accumulating safety data. Various regulatory mechanisms exist to support companies’ revised labeling to address new information. Once the need for an update is recognized, it is critical that submission to authorities occurs seamlessly due to the importance of such changes and the need for urgent implementation in some cases. Awareness of local requirements must be current to facilitate these updates and avoid local nuances that may delay the availability of important information to health care professionals and patients. Common pitfalls companies face include lack of awareness that changes cannot be submitted during ongoing license renewal processes in some countries, strict timelines for submission of safety changes or detailed dossier requirements (i.e., various countries including Egypt, India, Indonesia, Malaysia and Balkan countries specifically require clean and marked-up versions of the summary of product characteristics [SmPC] or comparative table against the previous version of the SmPC.) On the other hand, lack of awareness of beneficial regulatory processes may also result in unnecessary delays. For example, merging the safety/labeling update with an ongoing authority variation is possible in some countries and can result in faster approval.
Periodic safety update and benefit risk evaluation reports
Periodic safety update reports (PSURs), otherwise known as periodic benefit risk evaluation reports (PBRER), are pharmacovigilance documents that provide regulatory authorities with an update of the worldwide cumulative safety experience of a market-authorized product. The report must be periodically submitted to regulatory authorities on a pre-agreed timeline. Awareness of local regulatory requirements is fundamental to support PSUR submission planning, which should factor in the relevant reporting period, data lock point and the submission timelines. In some countries, delayed submission may lead to license cancellation and/or suspension. Some countries (e.g., India) require PSUR submission within 30 calendar days from the last day of the reporting period. In our experience, this timeline can be restrictive, particularly for companies used to following international standards that allow two to three months to finalize a PSUR. However, we’ve seen that awareness of local requirements can facilitate forward planning to support resource allocation and, if the timeline cannot be met, a well-timed waiver request may be accepted by the regulatory authority in many cases.
Renewals
In some countries, a license may need to be renewed by the issuing regulatory authority in order for it to remain valid. Once renewed, the license typically remains valid for a set time period or in some cases indefinitely, (e.g., Australia and Singapore). In some countries there is an option to apply for unlimited validity after the initial approval, (e.g., Serbia and Russia). This is also the case for most EU countries, although there are exceptions, where initial authorizations are typically valid for five years with one renewal application required, typically nine to 12 months before initial license expiry; following this first renewal, the license is usually valid indefinitely unless specified otherwise by the regulator. Regulatory authority dossier and processes also vary across countries in relation to renewing licenses. Some countries have significant renewal dossier requirements, (e.g., Albania and Serbia), whereas others have lesser requirements. In some countries it is not possible to submit changes to licenses when the license renewal application is ongoing. This may put market supply at risk and needs to be considered, particularly in the context of safety label updates, which may require urgent market awareness.
Minimize risks, maximize opportunities
The topics covered in this blog are not exhaustive but represent some common challenges companies face during post-authorization life cycle maintenance. As demonstrated here, awareness of country nuances through regulatory intelligence is fundamental to successful navigation of complicated and evolving regulations, and also aids in the benefit companies can derive from available supportive regulatory mechanisms. Importantly, a strong process for regulatory intelligence screening and management facilitates rapid change implementation for safety updates, ensuring regulatory compliance and making key information available to health care professionals and patients.