Observational Studies
Demonstrating product safety and value with innovative real-world study designs and delivery solutions
Observational (or non-interventional) studies play a significant role in demonstrating product safety, effectiveness and value for various stakeholders. Achieve both regulatory and market access success by aligning real-world evidence (RWE) generation with your clinical and commercial goals to deliver impactful evidence to the right audience at the right time.
Real-world observational studies collect and analyze data through both prospective and retrospective studies to inform development plans, gather patient insights, identify disease burden and unmet need, and meet post-authorization and post-marketing commitments.

Delivering quality with an integrated, strategic approach
Every product is different, with unique attributes and associated value. By applying a “think first” strategic approach, our integrated team of multi-disciplinary experts assesses every study to determine the right study design for the research need, providing smart, fit-for-purpose solutions to optimize study efficiencies, reduce research burden and improve evidence generation.
Whether you require a basic study design or a more innovative approach to evidence development, we partner with you to ensure the right study is used to generate the most meaningful results for your stakeholders.
Proactively anticipate and address questions and challenges at every step. Develop powerful, differentiating evidence to take your product further and reach your goals faster.
Informing decision-making with powerful evidence
Well designed and executed RWE studies have the power to answer countless questions and inform decision-making across all stages of product development and commercialization. Make the most of your real-world evidence with robust methods, tailored study designs, epidemiologic insights, therapeutic area expertise, advanced data analytics and precision study execution.
Augment and amplify your product’s value story with real-world evidence, addressing critical development questions, filling evidence gaps and monitoring real-world outcomes.
Identify unmet need and inform pipeline and research decisions
Gauge potential success and inform decisions on asset prioritization, feasibility, trial designs and product development strategy with studies such as:
- Natural history studies to understand disease insights on prevalence, demographics, changes in severity and patient outcomes
- Registry and biospecimen studies to identify target patient populations, predict clinical benefit and provide surrogate biomarkers for clinical trials
- External control arms to provide comparator data for single-arm studies, when randomized controlled trials (RCTs) are not sufficient or ethical
- Comparative effectiveness studies (cohort, case-control or cross-sectional studies) to assess the comparative effectiveness of treatments in the real world
- Modeling and simulation studies to simulate trial designs and outcomes and model economic impact of treatments
- Patient preference studies to design patient-centric studies that encourage recruitment and retention
Monitor real-world outcomes
Improve data-driven risk and safety assessment and management, measure safety and effectiveness of treatments in the real world, and meet post-approval requirements and commitments with studies such as:
- Post-market surveillance studies to identify adverse events that may not have appeared in clinical trials and support early safety detection and management
- Post-authorization safety studies (PASS) to assess the long-term safety profile of an intervention in real-world patient populations
- Post-authorization efficacy studies (PAES) to complement clinical efficacy data with a more comprehensive view of how well an intervention works in larger populations
- Risk evaluation and mitigation strategies (REMS) and risk management plans (RMPs) to identify and manage potential risks of marketed products and ensure an acceptable risk-benefit ratio
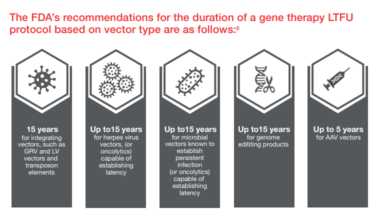
- Long-term follow-up (LTFU) studies to monitor the long-term effectiveness and safety of investigational and marketed products, especially important for cell and gene therapies, which require LTFU studies starting in Phase I
- Pregnancy and lactation studies to understand the safety and efficacy of interventions in these subpopulations and provide guidance on benefits and risks to ensure optimal care for pregnant individuals and their infants
Strengthening studies with diverse methodologies & operational excellence
Generating the best data for your needs requires a highly effective and efficient study design and excellent operational delivery. Through our integrated approach, we provide our clients with extensive methodological knowledge and experience, combined with established, global operational structure and processes.
Diverse data design approach
One methodology isn’t always the best solution when considering study design. Research goals are often best achieved by applying multiple data sources, including secondary (existing) data and primary data collection.

A strategic approach to study design
Understand multiple stakeholder evidence needs across the product life cycle and challenges they face.
Apply a “think first” approach to define a solution that aligns with a client’s strategy, and drive innovations to improve evidence and study effectiveness.
Recommend the optimal combination of standard and novel data sources to address the research question.
Optimize the use of available data, reduce primary data collection burden and deliver faster and less costly results with a diverse data design that considers all data options, including:
- Secondary RWD data sources (e.g., EMRs, chart reviews, claims, disease registries)
- RWD networks (e.g., integrated delivery networks, federated networks, health care organizations (HCOs))
- Social media
- Biosensors (e.g., blood pressure, blood glucose)
- Primary data collection (e.g., surveys, interviews)
- Patient-mediated data collection
Operational delivery excellence
Operational considerations are an essential aspect of deciding on the right study design to ensure superior delivery. Our highly qualified operational team brings implementation experience and solutions to elevate your study and achieve your goals. With access to the global footprint of Thermo Fisher Scientific, we provide clients with existing proven resources and processes, including:
- A global network of high-enrolling, established sites
- Technology platforms to streamline workflows and align to client preferences
- Decentralized study solutions
- Flexible data collection with less site and investigator burden
- Right-fit staffing models to meet study goals
- Experienced and tenured team members dedicated to delivery excellence
Exceed your study goals with a partner focused on assessing your specific research needs, providing tailored recommendations and delivering exceptional results.

Leveraging global experience
Our global team of scientific and operational experts, including clinicians, epidemiologists, data analysts, biostatisticians and data technologists, brings specialized knowledge to our integrated approach. Benefit from more than 30 years of experience and excellence to advance your product development.
- 35+ years of experience designing and operationalizing global, real-world studies
- 950+ RWE and market access professionals
- 270+ retrospective database studies (in the past five years)
- 380+ prospective, observational studies (in the past five years)
- Successful deployment of advanced technologies, such as artificial intelligence (AI), machine learning and natural language processing
- Methodological expertise and rigor to enhance the reliability of results
The addition of real-world studies to clinical trial data provides deeper and richer insights to demonstrate product safety, effectiveness and value. Strategically integrate observational research into your drug development plan for informed, evidence-based decisions at every stage of development to improve efficiencies, build stronger evidence and provide better access to life-changing interventions.
Relevant resources