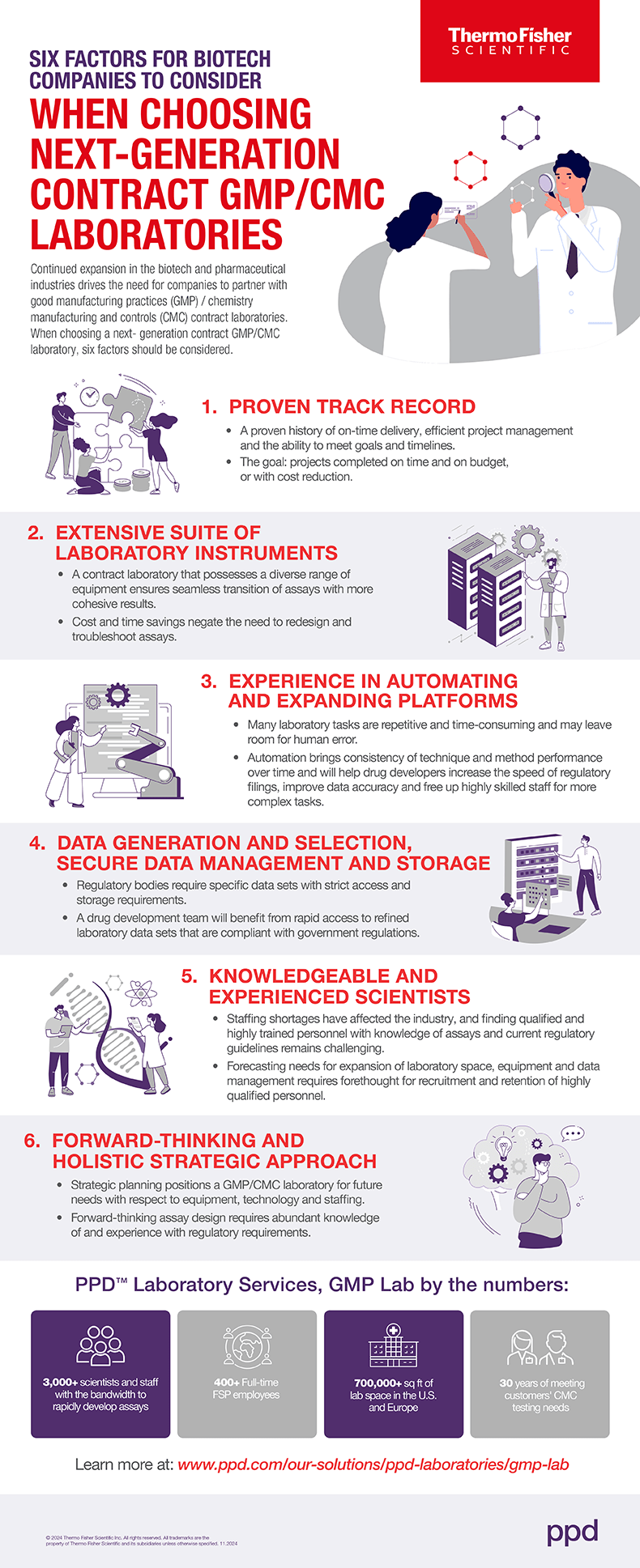
Six Factors for Biotech Companies to Consider When Choosing Next-Generation Contract GMP/CMC Laboratories
With continued expansion in biotech and pharma, these key considerations are critical when partnering with good GMP/CMC labs.

There has been an unprecedented expansion of different types of pharmaceutical therapies, as well as a change in the overall approach to health care. Continued expansion of the biotech industry is occurring in parallel, with an estimated compound annual growth rate of 13.96% from 2023-2030.1 With this growth comes increased demands for laboratory services at all steps across the development process.
As exponential growth and global expansion continue in both the biotech and pharmaceutical industries, it is critical for drug development companies to partner with good manufacturing practices (GMP) / chemistry manufacturing and controls (CMC) contract labs to conduct analyses of a range of small molecules and biologics and drive therapies forward efficiently.
Leading GMP/CMC contract labs will:
- Provide end-to-end support for drug developers across a range of products
- Share collective experience with access to highly trained scientists and the latest platforms, technologies and instruments
- Expedite processes through automation to provide substantial cost savings
- Reduce the time required to get a therapeutic to market
Successful assay development and validation require a significant investment of time and money, background knowledge of compliance with regulations, and for newer modalities, biosafety. Access to a laboratory partner that already possesses the equipment, knowledge and personnel can save time and money. For rapid expansion of late-stage assay platforms, speed and a proven track record are pivotal. However, an effective CRO doesn’t just perform assays; it provides comprehensive project planning, and also compiles, analyzes and packages data into an actionable and appropriate format for each individual client.
PPD® Laboratory services are backed by a proven track record with over three decades of experience providing CMC pharmaceutical testing. With the depth of knowledge to support all types of pharmaceutical products across all phases of development, we provide services ranging from end-to-end assay development to platform expansion, designed with the client’s timeline and budget in mind.